Collision Theory provides a qualitative explanation of chemical reactions and the rates at which they occur. A basic principal of collision theory is that, in order to react, molecules must collide. This fundamental rule guides any analysis of an ordinary reaction mechanism.
Consider the elementary bimolecular reaction:
If the two molecules A and B are to react, they must come into contact with sufficient force so that chemical bonds break. We call such an encounter a collision. If both A and B are gases, the frequency of collisions between A and B will be proportional to the concentration of each gas. If we double the concentration of A, the frequency of A-B collisions will double, and doubling the concentration of B will have the same effect. Therefore, according to collision theory, the rate at which molecules collide will have an impact on the overall reaction rate.
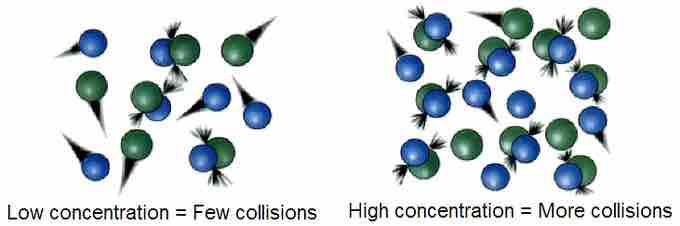
Molecular collisions
The more molecules present, the more collisions will happen.
Activation Energy and Temperature
When two billiard balls collide, they simply bounce off of one other. This is also the most likely outcome when two molecules, A and B, come into contact: they bounce off one another, completely unchanged and unaffected. In order for a collision to be successful by resulting in a chemical reaction, A and B must collide with sufficient energy to break chemical bonds. This is because in any chemical reaction, chemical bonds in the reactants are broken, and new bonds in the products are formed. Therefore, in order to effectively initiate a reaction, the reactants must be moving fast enough (with enough kinetic energy) so that they collide with sufficient force for bonds to break. This minimum energy with which molecules must be moving in order for a collision to result in a chemical reaction is known as the activation energy.
As we know from the kinetic theory of gases, the kinetic energy of a gas is directly proportional to temperature. As temperature increases, molecules gain energy and move faster and faster. Therefore, the greater the temperature, the higher the probability that molecules will be moving with the necessary activation energy for a reaction to occur upon collision.
Molecular Orientation and Effective Collisions
Even if two molecules collide with sufficient activation energy, there is no guarantee that the collision will be successful. In fact, the collision theory says that not every collision is successful, even if molecules are moving with enough energy. The reason for this is because molecules also need to collide with the right orientation, so that the proper atoms line up with one another, and bonds can break and re-form in the necessary fashion. For example, in the gas-phase reaction of dinitrogen oxide with nitric oxide, the oxygen end of N2O must hit the nitrogen end of NO; if either molecule is not lined up correctly, no reaction will occur upon their collision, regardless of how much energy they have. However, because molecules in the liquid and gas phase are in constant, random motion, there is always the probability that two molecules will collide in just the right way for them to react.
Of course, the more critical this orientational requirement is, like it is for larger or more complex molecules, the fewer collisions there will be that will be effective. An effective collision is defined as one in which molecules collide with sufficient energy and proper orientation, so that a reaction occurs.
Conclusion
According to the collision theory, the following criteria must be met in order for a chemical reaction to occur:
- Molecules must collide with sufficient energy, known as the activation energy, so that chemical bonds can break.
- Molecules must collide with the proper orientation.
- A collision that meets these two criteria, and that results in a chemical reaction, is known as a successful collision or an effective collision.