Structure of an Amino Acid
Amino acids are the monomers that make up proteins. Each amino acid has the same fundamental structure , which consists of a central carbon atom, also known as the alpha (α) carbon, bonded to an amino group (NH2), a carboxyl group (COOH), and to a hydrogen atom. In the aqueous environment of the cell, the both the amino group and the carboxyl group are ionized under physiological conditions, and so have the structures -NH3+ and -COO-, respectively. Every amino acid also has another atom or group of atoms bonded to the central atom known as the R group. This R group, or side chain, gives each amino acid proteins specific characteristics, including size, polarity, and pH.
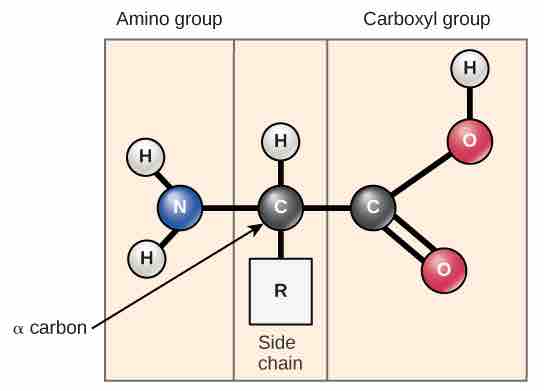
Amino acid structure
Amino acids have a central asymmetric carbon to which an amino group, a carboxyl group, a hydrogen atom, and a side chain (R group) are attached. This amino acid is unionized, but if it were placed in water at pH 7, its amino group would pick up another hydrogen and a positive charge, and the hydroxyl in its carboxyl group would lose and a hydrogen and gain a negative charge.
Types of Amino Acids
The name "amino acid" is derived from the amino group and carboxyl-acid-group in their basic structure. There are 21 amino acids present in proteins, each with a specific R group or side chain. Ten of these are considered essential amino acids in humans because the human body cannot produce them and they must be obtained from the diet. All organisms have different essential amino acids based on their physiology.
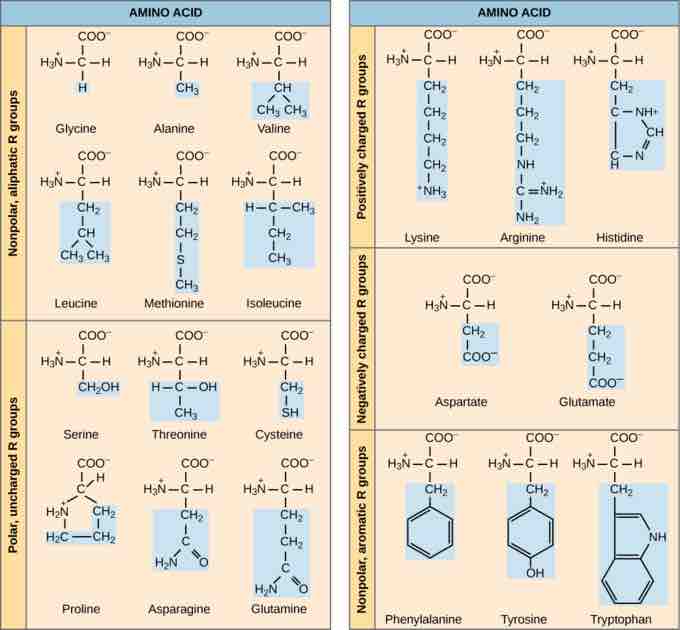
Types of amino acids
There are 21 common amino acids commonly found in proteins, each with a different R group (variant group) that determines its chemical nature. The 21st amino acid, not shown here, is selenocysteine, with an R group of -CH2-SeH.
Characteristics of Amino Acids
Which categories of amino acid would you expect to find on the surface of a soluble protein, and which would you expect to find in the interior? What distribution of amino acids would you expect to find in a protein embedded in a lipid bilayer?
The chemical composition of the side chain determines the characteristics of the amino acid. Amino acids such as valine, methionine, and alanine are nonpolar (hydrophobic), while amino acids such as serine, threonine, and cysteine are polar (hydrophilic). The side chains of lysine and arginine are positively charged so these amino acids are also known as basic (high pH) amino acids. Proline is an exception to the standard structure of an animo acid because its R group is linked to the amino group, forming a ring-like structure.
Amino acids are represented by a single upper case letter or a three-letter abbreviation. For example, valine is known by the letter V or the three-letter symbol val.
Peptide Bonds
The sequence and the number of amino acids ultimately determine the protein's shape, size, and function. Each amino acid is attached to another amino acid by a covalent bond, known as a peptide bond. When two amino acids are covalently attached by a peptide bond, the carboxyl group of one amino acid and the amino group of the incoming amino acid combine and release a molecule of water. Any reaction that combines two monomers in a reaction that generates H2O as one of the products is known as a dehydration reaction, so peptide bond formation is an example of a dehydration reaction.
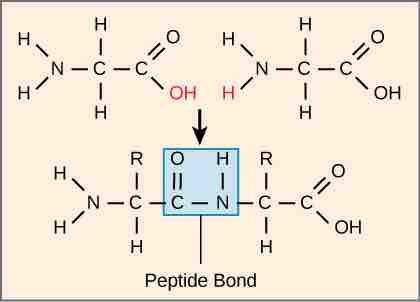
Peptide bond formation
Peptide bond formation is a dehydration synthesis reaction. The carboxyl group of one amino acid is linked to the amino group of the incoming amino acid. In the process, a molecule of water is released.
Polypeptide Chains
The resulting chain of amino acids is called a polypeptide chain. Each polypeptide has a free amino group at one end. This end is called the N terminal, or the amino terminal, and the other end has a free carboxyl group, also known as the C or carboxyl terminal. When reading or reporting the amino acid sequence of a protein or polypeptide, the convention is to use the N-to-C direction. That is, the first amino acid in the sequence is assumed to the be one at the N terminal and the last amino acid is assumed to be the one at the C terminal.
Although the terms polypeptide and protein are sometimes used interchangeably, a polypeptide is technically any polymer of amino acids, whereas the term protein is used for a polypeptide or polypeptides that have folded properly, combined with any additional components needed for proper functioning, and is now functional.