Bonding in Coordination Compounds
Many of the properties of metal complexes are dictated by their electronic structures. The electronic structure can be described by a relatively ionic model that ascribes formal charges to the metals and ligands. This approach is the essence of crystal field theory (CFT), which is a core concept in inorganic chemistry.
More sophisticated models (relative to crystal field theory) embrace covalency. This approach is described by the ligand field theory (LFT) and the molecular orbital theory (MO). Ligand field theory, introduced in 1935 and built from molecular orbital theory, can handle a broader range of complexes. It can explain complexes in which the interactions are covalent. The chemical applications of group theory can aid in the understanding of crystal or ligand field theory, by allowing simple, symmetry-based solutions to the formal equations.
Chemists tend to employ the simplest model required to predict the properties of interest. For this reason, CFT has been a favorite for the discussions when possible. MO and LF theories are more complicated but provide a more realistic perspective.
Color in Coordination Compounds
Metal complexes often have spectacular colors caused by electronic transitions due to the absorption of light. For this reason, they are often applied as pigments. Most transitions that are related to colored metal complexes are either d–d transitions or charge transfer bands. In a d–d transition, an electron in a d orbital on the metal is excited by a photon to another d orbital of higher energy. However, the electron remains centered on the metal.
Color of various Ni(II) complexes in aqueous solutions
From left to right, [Ni(NH3)6]2+, [Ni3]2+, [NiCl4]2-, [Ni(H2O)6]2+. Reactions starting from NiCl2·6H2O can be used to form a variety of nickel coordination complexes because the H2O ligands are rapidly displaced by ammonia, amines, thioethers, thiolates, and organophosphines.
A charge transfer band entails promotion of an electron from a metal-based orbital into an empty ligand-based orbital (Metal-to-Ligand Charge Transfer or MLCT). Conceptually, one can imagine the oxidation state of the metal increasing by one (losing on electron), while the oxidation state of the ligand decreases by one (becomes anionic). The overall charge of the system remains the same, but the localization of the electron changes.
The converse will also occur: excitation of an electron in a ligand-based orbital into an empty metal-based orbital (Ligand to Metal Charge Transfer or LMCT). These phenomena can be observed with the aid of electronic spectroscopy (also known as UV-Vis).
It is the relative energetics of these electronic transitions that allows for them to have absorbencies in the visible region. Since the nature of the ligands and the metal can be tuned extensively, a variety of colors can be obtained.
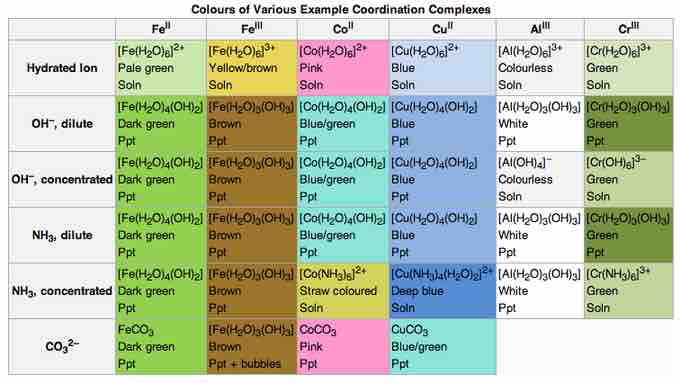
Colors of various coordination complexes
Changing the metal or the ligand can change the color of the coordination complex.