Concept
Version 10
Created by Boundless
Coordination Number, Ligands, and Geometries
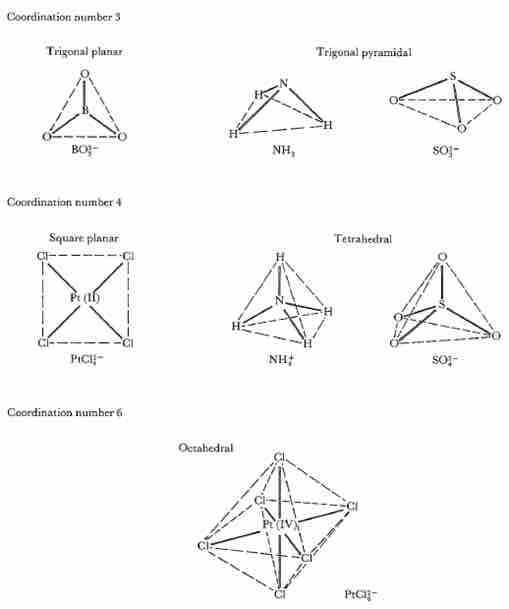
Geometry of atoms around central atoms with coordination numbers
Geometry of atoms around central atoms with coordination numbers 3, 4, and 6. If L is any peripheral atom and M is the central atom, then the bond angle L - M - L is 120° for trigonal planar, 109.5° for tetrahedral, and typically around 109.5° for trigonal pyramidal geometries. Square planar and octahedral geometries have two L - M - L angles, 90° and 180°.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
"Chemical Principles Fig 1.6."
http://en.wikibooks.org/wiki/File:Chemical_Principles_Fig_1.6.png
Wikibooks
CC BY-SA 3.0.