Concept
Version 10
Created by Boundless
Calculating Equilibrium Concentrations of Polyprotic Acids
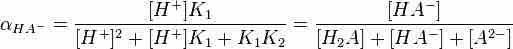
Equation for finding the fractional dissociation of HA-
The above concentration can be used if pH is known, as well as the two acid dissociation constants for each dissociation step; oftentimes, calculations can be simplified for polyprotic acids, however.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources: