Stereo Light Source
Interference microscopy uses a prism to split light into two slightly diverging beams that then pass through the specimen. It is thus based on measuring the differences in refractive index upon recombining the two beams. Interference occurs when a light beam is retarded or advanced relative to the other.
There are three types of interference microscopy: classical, differential contrast, and fluorescence contrast. Since its introduction in the late 1960s differential interference contrast microscopy (DIC) has been popular in biomedical research because it produces high-resolution images of fine structures by enhancing the contrasted interfaces. The image produced is of a thin optical section and appears three-dimensional, with a shadow around it. This creates a contrast across the specimen that is bright on one side and darker on the other.
The Interference Microscope
The microscope is a bright field light microscope with the addition of the following elements: a polarizer between the light source and the condenser, a DIC beam-splitting prism, a DIC beam-combining prism, and an analyzer . Manipulating the prism changes the beam separation, which alters the contrast of the image. When the two beams pass through the same material across the specimen they produce no interference. When the two beams pass through different material across the specimen such as on the edges, they produce alteration when combined.
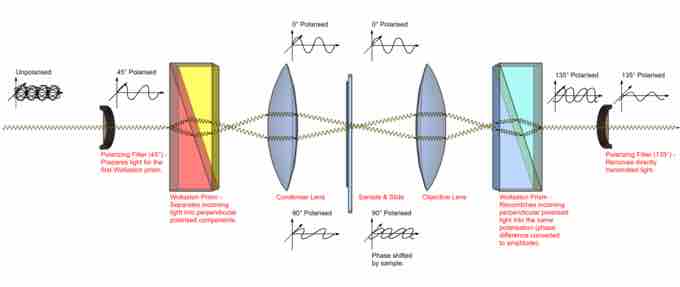
Path of light in differential interference contrast microscopy (DIC)
Two parallel light beams pass through the specimen and combine to produce an image.
Fluorescence differential interference contrast (FLIC) microscopy was developed by combining fluorescence microscopy with DIC to minimize the effects of photobleaching on fluorochromes bound to the stained specimen. The same microscope is equipped to simulataneously image a specimen using DIC and fluorescence illumination.