Concept
Version 11
Created by Boundless
Covalent Crystals
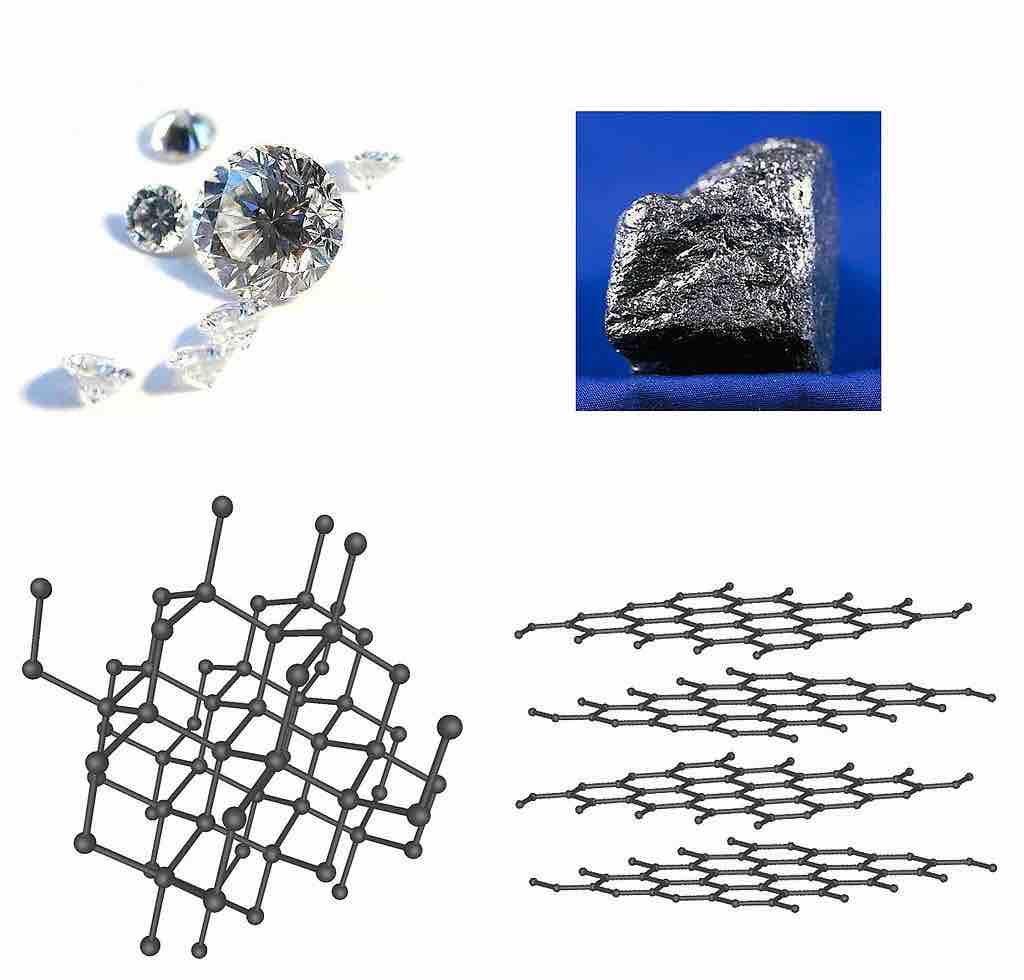
Diamond and Graphite: Two Allotropes of Carbon
These two allotropes of carbon are covalent network solids which differ in the bonding geometry of the carbon atoms. In diamond, the bonding occurs in the tetrahedral geometry, while in graphite the carbons bond with each other in the trigonal planar arrangement. This difference accounts for the drastically different appearance and properties of these two forms of carbon.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources:
"Diamond and graphite."
http://commons.wikimedia.org/wiki/File:Diamond_and_graphite.jpg
Wikimedia Commons
CC BY-SA 3.0.