Concept
Version 16
Created by Boundless
Hydrogen Bonding
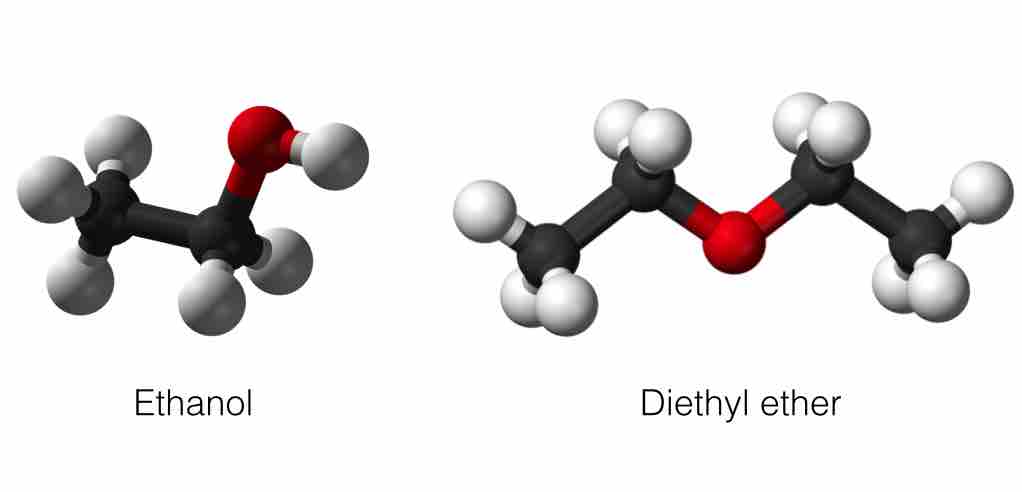
Hydrogen bond donor and hydrogen bond acceptor
Ethanol contains a hydrogen atom that is a hydrogen bond donor because it is bonded to an electronegative oxygen atom, which is very electronegative, so the hydrogen atom is slightly positive. Diethyl ether contains an oxygen atom that is a hydrogen bond acceptor because it is not bonded to a hydrogen atom and so is slightly negative.
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources: