Concept
Version 10
Created by Boundless
Bond Order
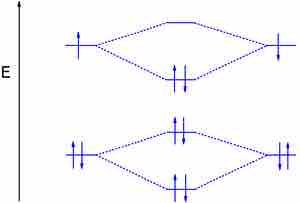
The dilithium molecule
Without the 1s electrons participating in bonding, the p electrons completely fill the bonding orbital; this leaves the antibonding orbital empty and gives a bond order of one, indicating a stable molecule (in this case, in the gas phase).
Source
Boundless vets and curates high-quality, openly licensed content from around the Internet. This particular resource used the following sources: